
We found that CDK2 signals colocalize with SUN1 and MLH1 signals in meiotic chromosomes in rats and mole voles, similar to the mouse. In the hamster, CDK2 signals were revealed in telomeric regions in the short synaptic segment of the sex bivalent. In female mole voles, the XX bivalent had CDK2 pattern similar to autosomes of all species. 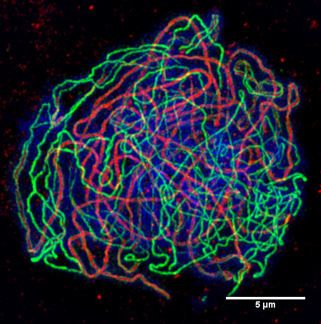
In the vole, sex chromosomes did not synapse, but demonstrated CDK2 signals of varying intensity, similar to the rat X and Y chromosomes. In the mole voles, which have unique XX sex chromosomes in males, CDK2 signals were nevertheless distributed similarly to the rat XY sex chromosomes. In the XY bivalent in the rat and mole rat, we detected numerous CDK2 signals in asynaptic regions and a single CDK2 focus on synaptic segments, similar to the mouse sex chromosomes. In sex bivalents following synaptic specificity, the CDK2 signals were distributed in three different modes. We showed that during prophase I, CDK2 localizes to telomeric and interstitial regions of autosomes in all species investigated (rat, vole, hamster, subterranean mole voles, and mole rats). Using an enhanced protocol, we were able to assess the distribution of as many as four separate proteins in the same meiotic cell. Antibodies to the following proteins were used: RAD51, a member of the double-stranded DNA break repair machinery MLH1, a component of the DNA mismatch repair system and SUN1, which is involved in the connection between the meiotic telomeres and nuclear envelope, alongside the synaptic protein SYCP3 and kinetochore marker CREST. To compare the distribution of CDK2 in XY and XX male sex chromosomes, we performed multi-round immunostaining of a number of marker proteins in meiotic chromosomes of the rat and subterranean mole voles. We used immunocytochemistry to study the localization of CDK2 during meiosis in seven rodent species that possess hetero- and homomorphic male sex chromosomes. The critical role of CDK2 in the progression of meiosis was demonstrated in a single mammalian species, the mouse. Researchers with the company mProbe and the Mayo Clinic have been conducting further testing of tissue samples collected during endoscopies to confirm and optimize the assay, and early data demonstrate the test's accuracy and reproducibility and show potential at identifying biomarkers that could serve as therapeutic targets for development of esophageal cancer treatments, ProPhase said.Cyclin-dependent kinases (CDKs) are crucial regulators of the eukaryotic cell cycle. ProPhase said study results published in 2018 suggest the BE-Smart test can detect protein expressions of cells at high risk of cancer development with 99 percent sensitivity and specificity. The company has set a reimbursement goal of $1,000-$3,000 per test. The transaction is expected to close in early January 2023, pending approval by Stella's shareholders.ĭoylestown, Pennsylvania-based ProPhase plans to validate the BE-Smart mass spec-based proteomic assay as a laboratory-developed test, and the company predicts it will commercialize the test within 18 months. Stella will receive another $2 million of ProPhase common stock upon achievement of a revenue-based commercial milestone within five years after closing, and will receive a 5 percent royalty on the adjusted gross margin from commercialization of the intellectual property. Under the terms of the agreement, Salt Lake City-based Stella will receive $3.5 million in cash and $1 million in ProPhase common stock. NEW YORK – ProPhase Labs said Monday it had agreed to a $4.5 million deal to buy the exclusive rights to Stella Diagnostics' BE-Smart Esophageal Pre-Cancer screening test and related intellectual property assets.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
